Electrostatic Force
Electrostatic Force formula |
||
|
\( F \;=\; k_e \cdot \dfrac{ | q_1 \cdot q_2 | }{ r^2 }\) (Electrostatic Force) \( k_e \;=\; \dfrac{ F \cdot r^2 }{ q_1 \cdot q_2 } \) \( q_1 \;=\; \dfrac{ F \cdot r^2 }{ k_e \cdot q_2 } \) \( q_2 \;=\; \dfrac{ F \cdot r^2 }{ k_e \cdot q_1 } \) \( r \;=\; \dfrac{ k_e \cdot q_1 \cdot q_2 }{ F } \) |
||
| Symbol | English | Metric |
| \( F \) = Electrostatic Force | \(lbf\) | \(N\) |
| \( k_e \) = Coulomb's Constant | \(lbf-ft^2 \;/\; F^2\) | \(N-m^2 \;/\; C^2\) |
| \( q_1, q_2 \) = Magnitudes of Two Electric Charges | \(C\) | \(C\) |
| \( r \) = Distance Between the Centers of the Two Charges | \(ft\) | \(m\) |
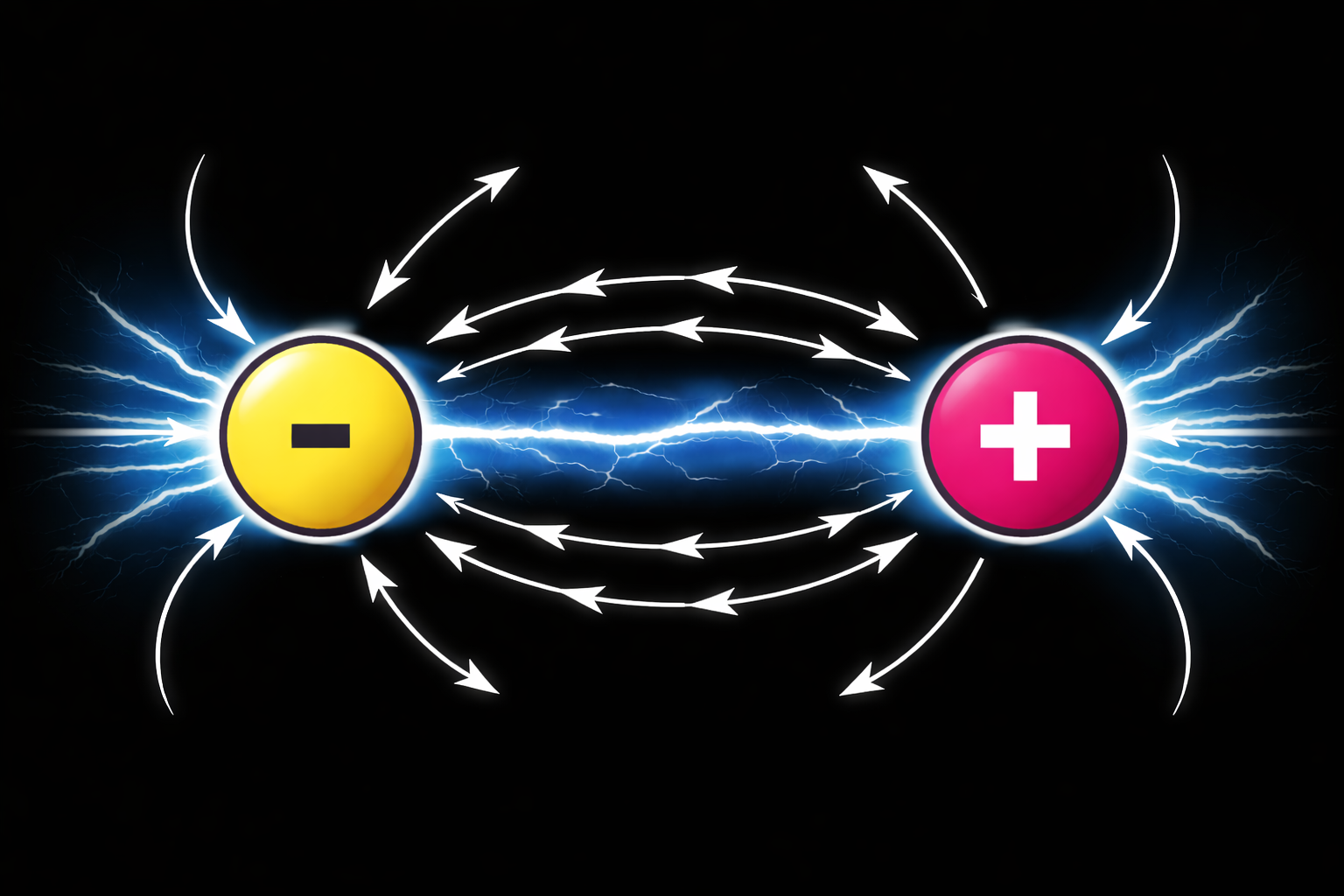
Electrostatic force, abbreviated as \(F\), is the force of attraction or repulsion that occurs between two charged particles or objects due to their electric charges. It arises from the fundamental property of matter known as electric charge, which can be either positive or negative. Like charges repel each other, while unlike charges attract, and this interaction takes place even without direct contact, acting over a distance through the electric field surrounding each charge. The magnitude of the electrostatic force depends on the amount of charge on the objects and the distance between them, as described by Coulomb’s law. This force plays a role in many natural and technological processes, influencing atomic and molecular interactions, material behavior, and the operation of electrical devices.

