Superheated Vapor
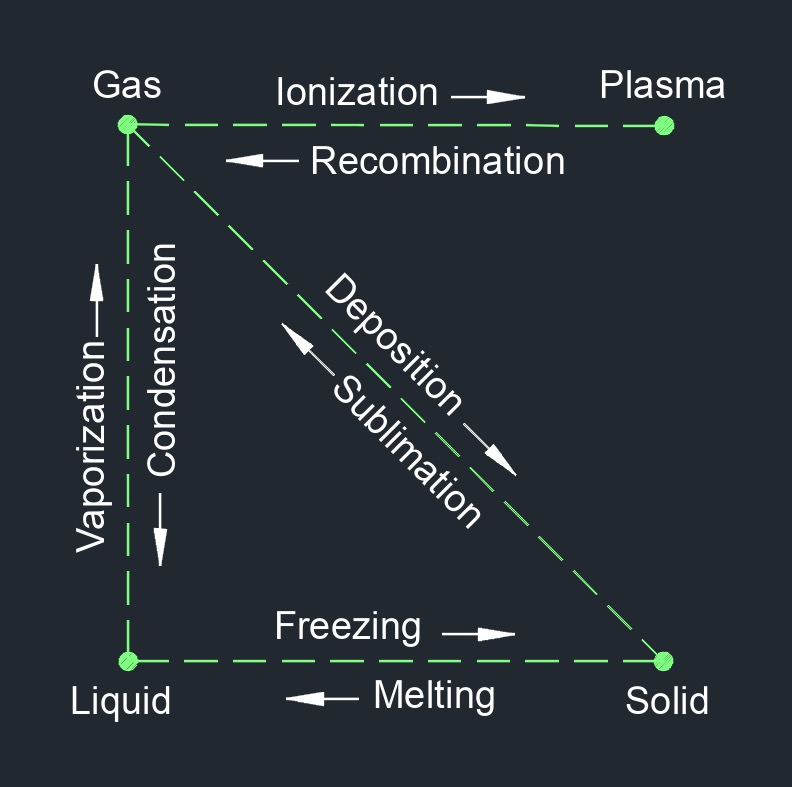
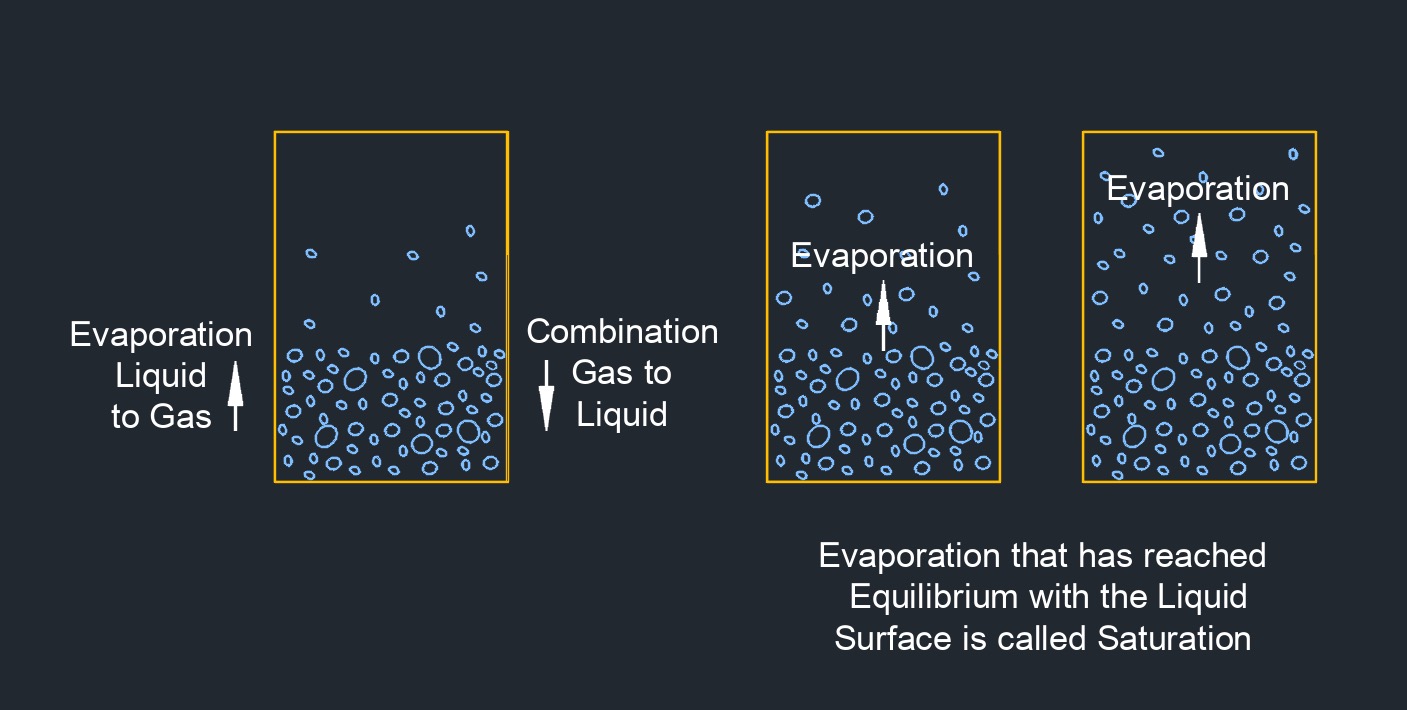 Superheated vapor, abbrevated as \(SHV\), is a vapor or gas that is in a state where its temperature exceeds the saturation temperature at a given pressure. It is a phase of a substance, such as water or any other material, in which the vapor exists at a higher temperature than what would be expected at the corresponding pressure. When a substance undergoes a phase change from liquid to gas, it initially becomes saturated vapor. However, if additional heat is supplied to the saturated vapor while maintaining the pressure constant, its temperature will rise beyond the saturation temperature, resulting in superheated vapor.
Superheated vapor, abbrevated as \(SHV\), is a vapor or gas that is in a state where its temperature exceeds the saturation temperature at a given pressure. It is a phase of a substance, such as water or any other material, in which the vapor exists at a higher temperature than what would be expected at the corresponding pressure. When a substance undergoes a phase change from liquid to gas, it initially becomes saturated vapor. However, if additional heat is supplied to the saturated vapor while maintaining the pressure constant, its temperature will rise beyond the saturation temperature, resulting in superheated vapor.
Superheated vapor is commonly used in power plants to drive steam turbines. By superheating the vapor, its energy content is increased, leading to improved energy conversion efficiency in the turbine.
In industrial processes, superheated vapor may be employed in applications that require high temperature heat transfer or when the absence of liquid water is necessary to prevent issues or unwanted reactions. It is worth noting that handling superheated vapor requires caution due to its high temperature. operation and prevent hazards.

