Petrochemicals are produced through refining and chemical processing operations that separate and transform hydrocarbons found in crude oil and natural gas. The primary raw hydrocarbons include compounds such as methane, ethane, propane, butane, naphtha, and other fractions obtained during petroleum refining. Through processes such as cracking, reforming, polymerization, and synthesis, these hydrocarbons are converted into simpler chemical building blocks. Among the most important petrochemical base compounds are ethylene, propylene, benzene, toluene, xylene, and methanol. These substances are foundational materials in modern chemical manufacturing.
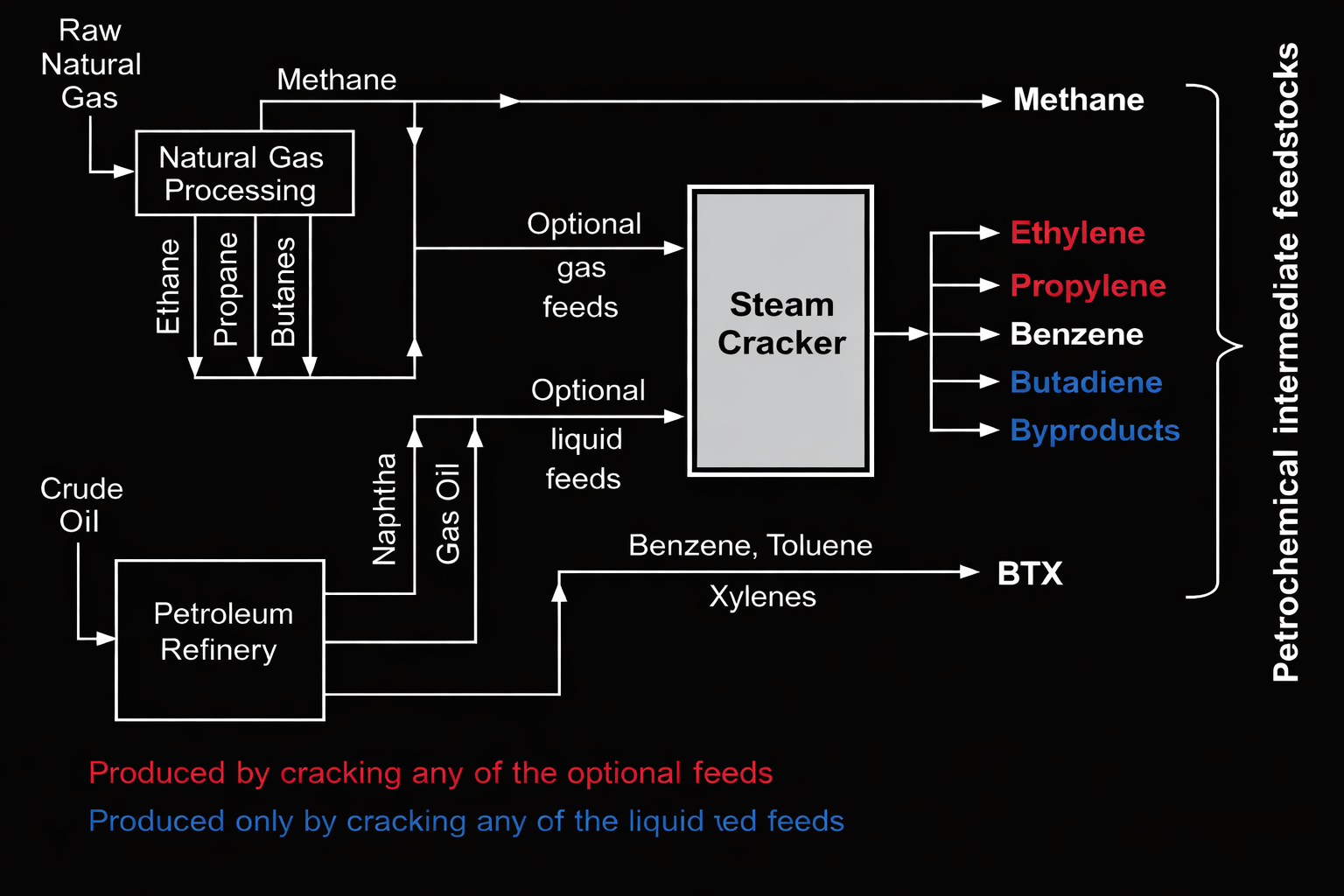
Aromatics - These primarily include benzene, toluene, and xylenes (collectively BTX). They are typically obtained from petroleum refineries via catalytic reforming of naphtha and are used in the production of dyes, detergents, polyurethanes, solvents, plastics, and synthetic fibers.
Olefins (Alkenes) - These include ethylene, propylene, butenes, and butadiene. They are produced mainly via steam cracking of hydrocarbons and serve as building blocks for plastics (e.g., polyethylene, polypropylene), synthetic rubbers, and many industrial chemicals.
Synthesis Gas (Syngas) and Related - This includes mixtures of carbon monoxide and hydrogen (often with methane derivatives), used to produce methanol, ammonia (for fertilizers like urea), and other chemicals. Some classifications also group C1 compounds like methane separately.
Petrochemicals are further processed into derivatives and end products (e.g., polymers/plastics, fibers, rubbers, solvents, fertilizers), but these are downstream applications rather than core branches of the petrochemicals themselves.
 Petrochemical Processes
Petrochemical Processes
Alkylation - Combines light olefins such as propylene or butylene with isobutane in the presence of strong acid catalysts. The process produces high-octane hydrocarbons used in gasoline blending.
Ammonia Production - Production commonly uses the Haber–Bosch process. Hydrogen, often derived from natural gas through steam methane reforming, reacts with nitrogen under high pressure and temperature in the presence of iron-based catalysts to form ammonia.
Aromatics Production - Processes isolate or generate benzene, toluene, and xylene from reformate streams or pyrolysis gasoline. These compounds are essential feedstocks for synthetic fibers, detergents, resins, plastics, and pharmaceuticals.
Catalytic Cracking - Converts heavy petroleum fractions into lighter hydrocarbons such as gasoline-range compounds, olefins, and liquefied petroleum gases. A solid acid catalyst is used to promote molecular bond breaking at lower temperatures than thermal cracking.
Catalytic Reforming - Reforming converts low-octane naphtha into high-octane aromatic-rich products using platinum-based catalysts. The process produces aromatic hydrocarbons such as benzene, toluene, and xylene, collectively known as BTX aromatics, along with hydrogen gas.
Chlorination - Introduces chlorine atoms into hydrocarbons or organic compounds to produce substances such as vinyl chloride monomer, which is used to manufacture polyvinyl chloride (PVC).
Coking - Delayed coking and related coking processes thermally decompose heavy residual oils into lighter hydrocarbons and solid petroleum coke.
Condensation Processes - Widely used in producing resins, synthetic fibers, and engineering plastics such as polyesters and polyamides.
Cumene Process - Reacts benzene with propylene to form cumene, which is then oxidized and cleaved to produce phenol and acetone.
Dehydrogenation - Removes hydrogen from hydrocarbons to produce olefins. For example, propane dehydrogenation produces propylene, and butane dehydrogenation produces butadiene.
Desulfurization - Remove sulfur compounds from feedstocks and products to reduce corrosion, catalyst poisoning, and sulfur emissions.
Esterification - Reacts acids and alcohols to form esters, which are used in solvents, polymers, lubricants, fragrances, and plasticizers.
Ethylene Oxide and Ethylene Glycol Production - Ethylene is oxidized to ethylene oxide, which is then hydrolyzed to ethylene glycol. Ethylene glycol is widely used in polyester production and antifreeze formulations.
Fischer–Tropsch Synthesis - Converts synthesis gas into liquid hydrocarbons using metal catalysts such as iron or cobalt. The process is used in gas-to-liquids and coal-to-liquids technologies.
Gasification - Converts hydrocarbons, coal, petroleum coke, or biomass into synthesis gas (syngas), mainly carbon monoxide and hydrogen, through partial oxidation at high temperatures. Syngas is used for producing methanol, ammonia, hydrogen, and synthetic fuels.
Hydrocracking - Combines catalytic cracking with hydrogenation. Heavy hydrocarbons are reacted with hydrogen under high pressure and temperature in the presence of catalysts to produce lighter and cleaner products such as diesel fuel, jet fuel, naphtha, and feedstocks for petrochemicals.
Hydrogenation - Adds hydrogen to unsaturated hydrocarbons or chemical intermediates. The process is widely used to saturate double bonds, stabilize products, and manufacture numerous petrochemicals.
Hydrotreating - Removes sulfur, nitrogen, oxygen, metals, and other impurities from petroleum fractions by reacting them with hydrogen over catalysts. This process is important for producing cleaner fuels and preparing feedstocks for downstream petrochemical operations.
Isomerization - Rearranges hydrocarbon molecules into different structural forms without changing their molecular formula. Straight-chain hydrocarbons are converted into branched isomers to improve fuel quality and octane rating.
Methanol Synthesis - Reacts carbon monoxide, carbon dioxide, and hydrogen over catalysts under elevated temperature and pressure to produce methanol. Methanol is an important petrochemical feedstock and fuel component.
Nitration and Sulfonation - Certain petrochemical intermediates undergo nitration or sulfonation to produce dyes, detergents, explosives intermediates, surfactants, and specialty chemicals.
Oxidation Processes - Controlled oxidation reactions produce chemicals such as ethylene oxide, propylene oxide, acetic acid, formaldehyde, and phthalic anhydride. These are important intermediates in plastics, solvents, and synthetic materials manufacturing.
Oxo Process (Hydroformylation) - Reacts olefins with carbon monoxide and hydrogen to produce aldehydes, which are later converted into alcohols, plasticizers, detergents, and solvents.
Polymerization - Processes chemically combine small molecules called monomers into large-chain polymers. Ethylene can be polymerized into polyethylene, propylene into polypropylene, and styrene into polystyrene. Polymerization methods include addition polymerization, condensation polymerization, bulk polymerization, suspension polymerization, and emulsion polymerization.
Pyrolysis - Pyrolysis thermally decomposes hydrocarbons in the absence of oxygen to produce smaller molecules, gases, liquids, and carbon-rich solids. Steam cracking is a specialized form of pyrolysis.
Separation and Purification Processes - Petrochemical plants rely heavily on physical separation methods such as distillation, fractional distillation, extraction, absorption, adsorption, stripping, crystallization, and membrane separation. These processes isolate valuable chemical products from complex hydrocarbon mixtures.
Steam Cracking - One of the most important petrochemical processes. Hydrocarbon feedstocks such as ethane, propane, butane, naphtha, or gas oil are heated at very high temperatures, typically about 750–900 °C, in the presence of steam. The process breaks large hydrocarbon molecules into smaller unsaturated hydrocarbons called olefins, primarily ethylene, propylene, and butadiene. Ethylene and propylene are major building blocks for plastics, synthetic fibers, solvents, and other chemicals.
Steam Methane Reforming (SMR) - Reforming reacts methane with steam at high temperatures over catalysts to produce hydrogen and carbon monoxide. It is the dominant industrial process for hydrogen production.
Vinyl Chloride Production - Vinyl chloride monomer is typically produced from ethylene through chlorination and cracking reactions and is used to manufacture PVC plastics.

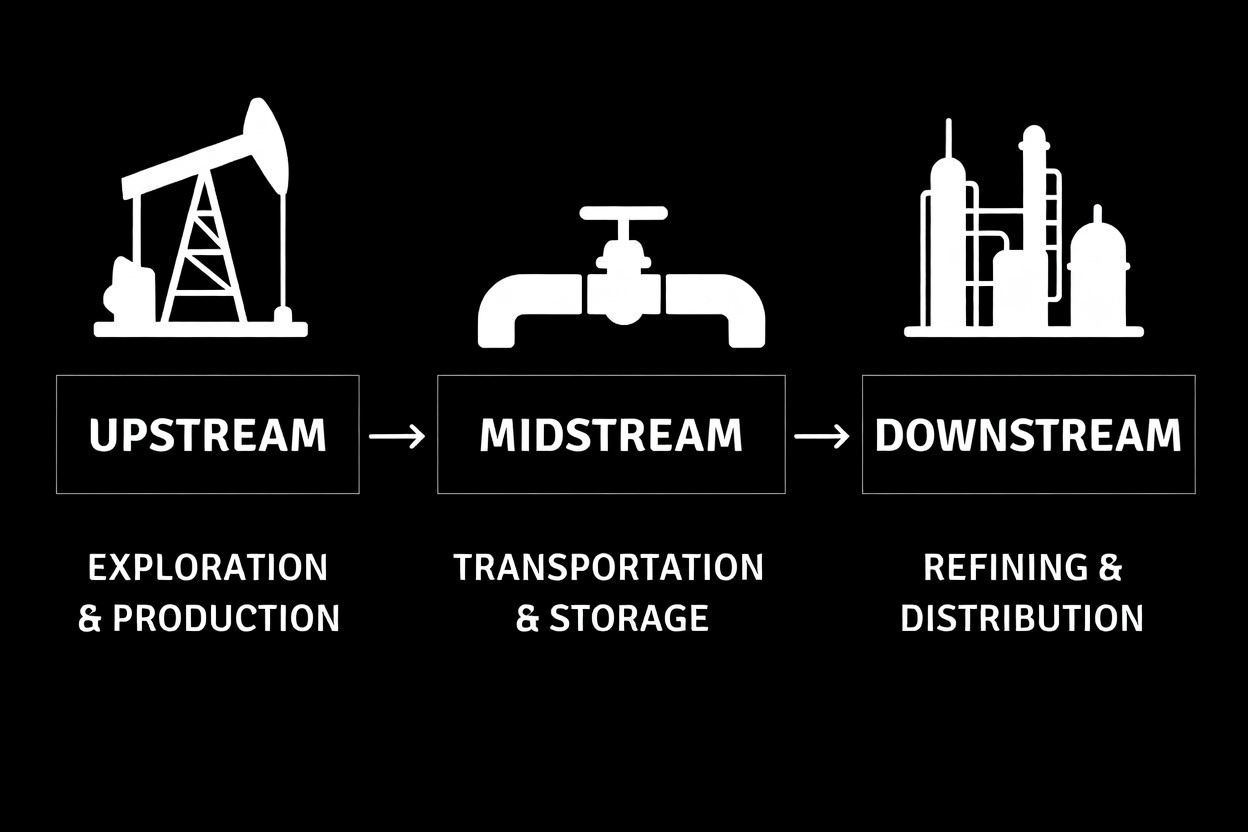 Petrochemical is a chemical substance that is derived from petroleum or natural gas. Petrochemicals are not usually used directly as fuels; instead, they serve as raw materials, also called feedstocks, for manufacturing a very large range of industrial and consumer products.
Petrochemical is a chemical substance that is derived from petroleum or natural gas. Petrochemicals are not usually used directly as fuels; instead, they serve as raw materials, also called feedstocks, for manufacturing a very large range of industrial and consumer products. Petrochemical Processes
Petrochemical Processes
