Specific Heat Capacity
Specific heat capacity, abbreviated as \(Q\), often simply referred to as specific heat, is a property of a substance that quantifies how much heat energy is required to raise the temperature of a unit mass of that substance by one degree Celsius (or one Kelvin). Or you could say, the amount of heat or energy required to increase the temperature of a substance by 1°C. It represents the substance's ability to store thermal energy.
Specific Heat Capacity Formula |
||
|
\( Q \;=\; c \cdot m \cdot \Delta T \) (Specific Heat Capacity) \( c \;=\; \dfrac{ Q }{ m \cdot \Delta T } \) \( m \;=\; \dfrac{ Q }{ c \cdot \Delta T } \) \( \Delta T \;=\; \dfrac{ Q }{ c \cdot m } \) |
||
| Symbol | English | Metric |
| \( Q \) = Specific Heat Capacity | \(Btu\;/\;lbm-F\) | \(kJ\;/\;kg-K\) |
| \( c \) = Specific Heat | \(Btu\;/\;lbm-F\) | \(kJ\;/\;kg-K\) |
| \( m \) = Mass | \(lbm\) | \(kg\) |
| \( \Delta T \) = Change in Temperature | \(^\circ F\) | \(^\circ K\) |
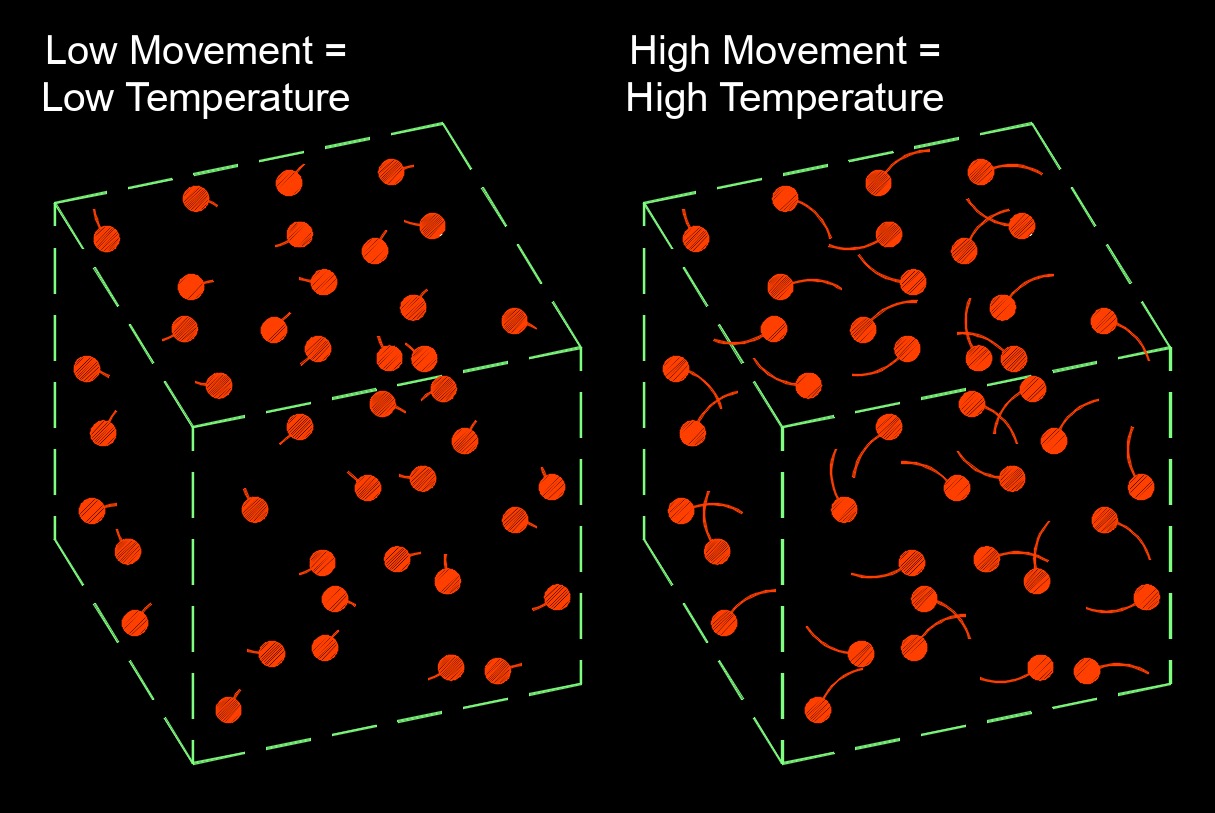 Different substances have different specific heat capacities due to their molecular and atomic structures. For example, water has a relatively high specific heat capacity, which means it can absorb or release a significant amount of heat energy without undergoing large temperature changes. This property makes water important for moderating temperature changes in the environment and is why it is used in cooling systems and as a reference for specific heat capacity.
Different substances have different specific heat capacities due to their molecular and atomic structures. For example, water has a relatively high specific heat capacity, which means it can absorb or release a significant amount of heat energy without undergoing large temperature changes. This property makes water important for moderating temperature changes in the environment and is why it is used in cooling systems and as a reference for specific heat capacity.
The specific heat capacity is an important property in thermodynamics and heat transfer calculations. It determines how much energy is needed to heat or cool a substance and is used in designing heating and cooling systems, understanding heat transfer processes, and analyzing the thermal behavior of materials.

